I chose to discuss the importance of pH balance in the blood as it relates to enzymatic function, cellular respiration as a cause of lowered pH and buffers in the blood to help keep pH in the optimum range by drawing cartoons on a pizza box.


I chose to discuss the importance of pH balance in the blood as it relates to enzymatic function, cellular respiration as a cause of lowered pH and buffers in the blood to help keep pH in the optimum range by drawing cartoons on a pizza box.
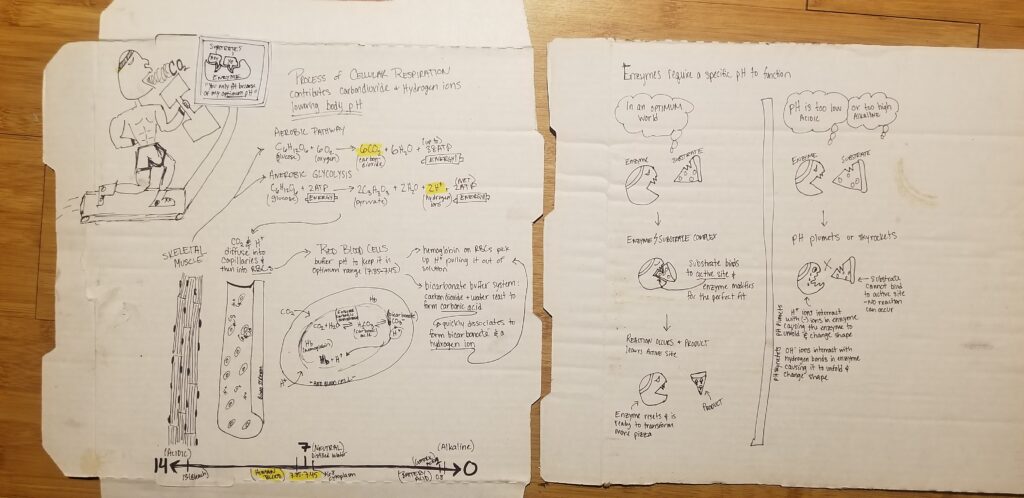
The Importance of an optimum pH balance in the human body is illustrated by Karen Shuster in cartoon form. A balanced pH is important in the body as it helps to maintain the mental state, muscle function, and respiratory drive. The scale ranging 0-14 pH, 7 being the neutral level of both, is measured by the type of ions released, as acids release Hydrogen ions and bases release Hydroxyl ions. An optimum pH balance is created when an enzyme and substrate bind in the active site. In the active site, the enzyme ensures the substrate is the right product makeup by modifying it. The bonded enzyme and substrate become one product that leaves the active site and the enzyme returns to its natural state to create more products again. If the pH is too low it can become acidic, but if it is too high it can be alkaline. If that occurs, the Hydrogen ions in the enzymes change shape and the substrate isn’t allowed to bind in the active site which in turn keeps a reaction from occurring. In another case, the OH ions in the enzyme react to the hydrogen bonds which causes them to change shape. Cellular Respiration is a big factor in the lowering of the pH by the use of carbon dioxide and Hydrogen ions. The R-groups of the Enzymes dictate where each substrate is meant to be depending on if they are polar, non-polar, acidic, alkaline, big or small, positive or negative, and neutral. A faster rate of reacting enzymes and substrates causes the ionization of both to have a hard time binding to each other. Whereas, a decrease in pH of the area the higher rate of Hydrogen ions that interact with negative charges of the enzymes causing the enzymes to change shape and stops them from binding to each other.