Hemoglobin is a protein found in red blood cells that binds to oxygen and transports it throughout the body. Why does it bind to oxygen though? Are there other molecules that it may bind to instead? Carbon monoxide is a chemical commonly produced by incomplete combustion and exists in small quantities in normal atmosphere (NIOSH, 2018).
Hemoglobin is a critical component of our blood and allows red blood cells to perform their major function of oxygen distribution. A normal hemoglobin level for a male is 14-18g/dl and for females is 12-16g/dl (Billett, 1990). Hemoglobin is made up of four molecules, broken up into two dimers of an alpha and beta chain combined. Each molecule contains an iron containing “heme” group that binds to oxygen, meaning each molecule of hemoglobin can carry four oxygen molecules. Hemoglobin, due to mutation, has many recognized alternative forms. Some forms function normally and are able to transport oxygen adequately or even better than normal, Hb F, while some have adverse or deleterious effects to the transport of oxygen including the form found in sickle cell anemia. The primary form, Hemoglobin A, accounts for 97% of hemoglobin in a “normal” individual (Billett, 1990).
Hemoglobin has four oxygen binding sites as mentioned previously. These sites are “heme” groups and are planar molecules that center an Fe2+ ion (Jiang et al., 2024).. Oxygen binds to these groups creating an octahedral iron complex called oxyhemoglobin (Schaller, n.d.). Hemoglobin binding no oxygen is called deoxyhemoglobin and binding carbon monoxide is called carboxyhemoglobin. Hemoglobin is useful because it can both bind oxygen tightly under the right conditions but also let go under the right conditions, following Bohr’s Law (Schaller, n.d)
Carbon monoxide has 300 times the binding affinity for hemoglobin iron groups than oxygen does (NIOSH, 2018). Carbon monoxide toxicity is an emergency that first responders must be aware of and identify quickly throughout their multitude of calls. A source of carbon monoxide in an enclosed space is not uncommon, especially during emergency situations that knock out power for long periods of times such as hurricanes, tornadoes, blizzards, and earthquakes. A hemoglobin that encounters CO will form carboxyhemoglobin, a more stable complex than oxyhemoglobin (Jiang et al., 2024). How are we able to treat patients that have been exposed to too much carbon monoxide?
Carbon monoxide toxicity can be treated through modern means. Hyperbaric chambers with increased oxygen is the most common method. Increased pressure in these chambers increases the amount of oxygen in the air and in the lungs. This increased concentration of oxygen affects the bond carbon monoxide has with the hemoglobin molecule, weakening its bond strength by as much as 50% (Jiang et al., 2024). This weakened bond can be overcome by hemoglobin’s affinity for oxygen, thus changing carboxyhemoglobin into oxyhemoglobin.
This finding can help create new treatments for carbon monoxide toxicity that may not require hyperbaric treatment. Medicine is ever changing and ever developing. Findings such as these may inspire the creation of medications and techniques that more easily save and extend life and more research will only expand these fields in previously unknown ways.
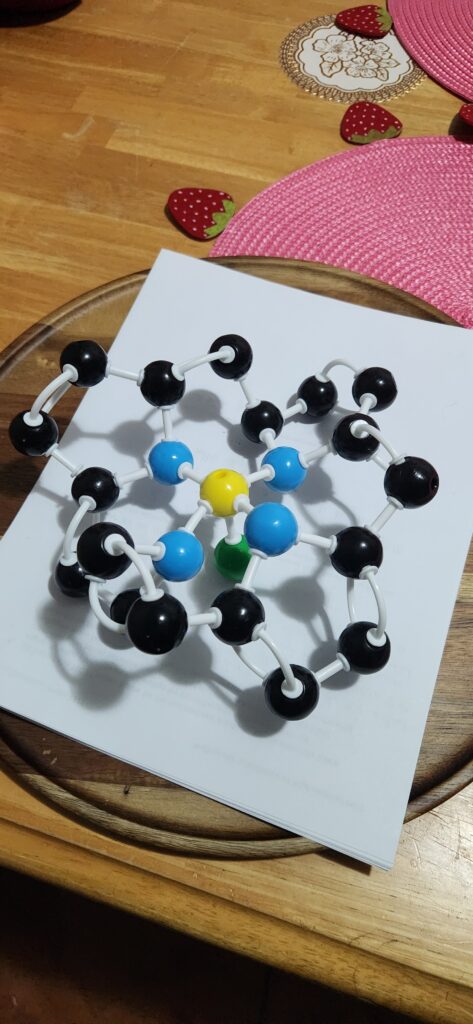
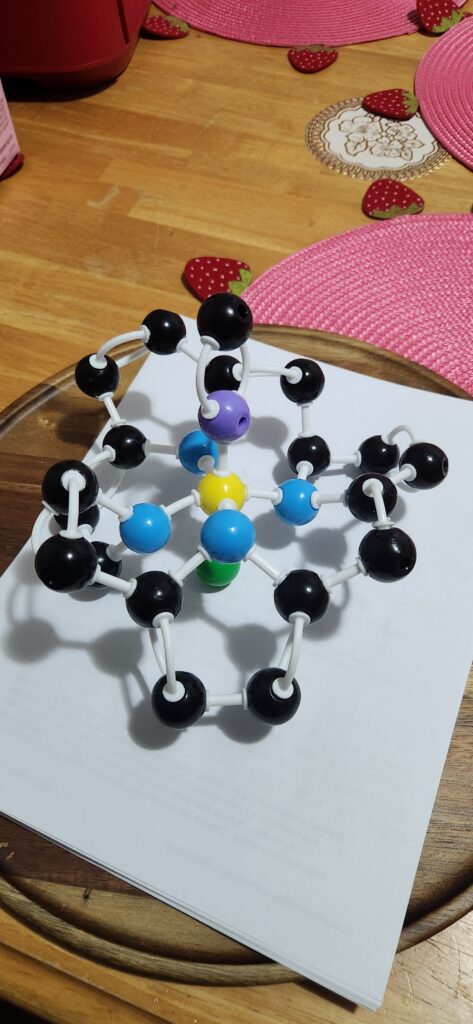
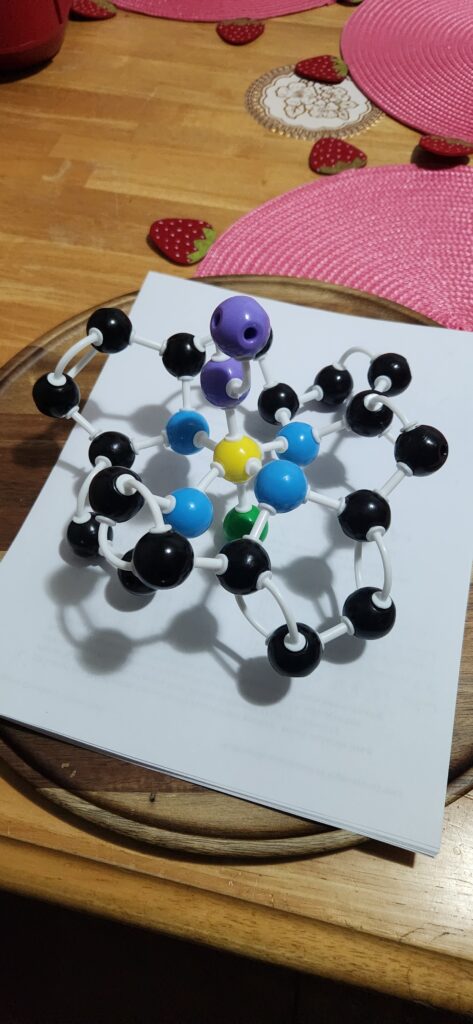
Guide to atoms:
Black: Carbon
Blue: Nitrogen
Yellow: Iron
Green: Continued molecule
Deoxyhemoglobin
Carboxyhemoglobin
Oxyhemoglobin
References
Jiang, M., Yu, C.-H., Xu, Z., & Qin, Z. (2024). Binding of Carbon Monoxide to Hemoglobin in an Oxygen Environment: Force Field Development for Molecular Dynamics. American Chemical Society. 10.1021
National Institute for Occupational Safety and Health. (2018). Carbon Monoxide. NIOSH. https://www.cdc.gov/niosh/topics/co-comp/default.html
Schaller, C. P. (n.d.). 4.2: Oxygen Binding. Chem.Libretexts.org. Retrieved April 19, 2024, from https://chem.libretexts.org/Bookshelves/General_Chemistry/Book%3A_Structure_and_Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_(Schaller)/V%3A__Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_3/04%3A_Oxygen_Binding_and_Reduction/4.02%3A_
Walker, H., Hall, W., & Hurst, J. (1990). Hemoglobin and Hematocrit – Clinical Methods. NCBI. Retrieved April 19, 2024, from https://www.ncbi.nlm.nih.gov/books/NBK259/
In Progress Photos



STEM Abstract
My STEM abstract is on Hemoglobin and its binding to affinity. Hemoglobin is a protein containing iron that facilitates the transport of oxygen in red blood cells. Hemoglobin in the blood carries oxygen flow from the respiratory organs such as the lungs to the body’s other tissues, where it releases the oxygen to enable aerobic respiration. Hemoglobin exists in 2 forms, the taut form (T) and the relaxed form (R). This structural change leads to low-affinity hemoglobin, whereas the relaxed form leads to a high-affinity form of hemoglobin to oxygen binding.
Carbon Monoxide increases hemoglobin binding affinity. The binding of one CO molecule to hemoglobin increases the affinity of the other binding spots for oxygen. This shift prevents oxygen unloading in peripheral tissue and therefore the oxygen concentration. The tissue is much lower than normal. (Patel, Jose, Mohiuddin) Carbon monoxide has 300 times the binding affinity for hemoglobin iron groups than oxygen does. Hemoglobin that encounters CO will form carboxyhemoglobin, a more stable complex than oxyhemoglobin.
The presence of carbon monoxide in a person can experience severe tissue, hypoxia while maintaining a normal PaO2. (Patel, Jose, Mohiuddin) Patients with CO poisoning experience symptoms such as headaches, malaise, altered mental status, shortness of breath, seizures, or cherry-red lips. (Patel, Jose. Mohiuddin) A pulse oximeter would usually be normal because the machine is unable to detect carboxyhemoglobin from oxyhemoglobin. This finding can help with creating new treatments that may not require hyperbaric treatments.