Ethan Griswold – STEAM Project
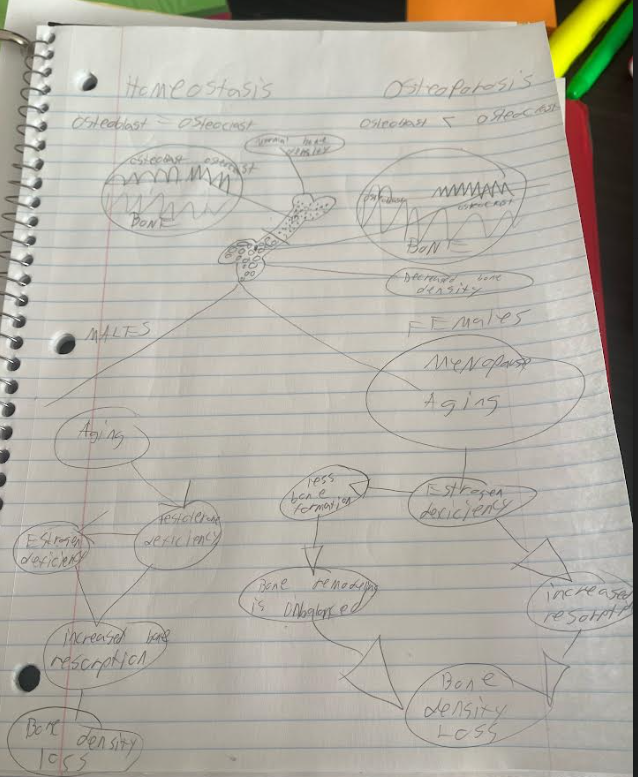
My project is on the role sex hormones play on bone disease, specifically osteoporosis. It looks at how our hormones fluctuate with age, and how they influence skeletal homeostasis on the cellular level. It then takes a brief look at intervention and prevention of osteoporosis. My project covers course the course objective “explain how bone development is hormonally regulated.” The basic idea of my “art” was to show the difference between a normal bone and a bone with osteoporosis, as well as what drives osteoporosis in men and women. As you can see, our hormones, estrogen and testosterone, play a big part in developing osteoporosis, which results in lower bone density.
Bone development commences in utero, where a template for the future skeleton is established through endochondral ossification (Almeida et al., 2017). During this period, the developing fetus relies on hormonal interaction for proper skeletal formation. As growth progresses, sex hormones begin to exert their influence. During childhood, growth hormone stimulates the proliferation of chondrocytes in the growth plates, contributing to longitudinal bone growth (Almeida et al., 2017). Sex hormones become more pronounced during puberty. Puberty unleashes a surge of sex hormones, signaling the closure of the growth plates and the consolidation of bone density. This will allow peak bone mass to be developed by our late 20s. As age progresses, a decline in sex hormones will ultimately lead to more bone being broken down, making it essential to achieve the most bone mass we can while still young.
Osteoporosis, a condition characterized by reduced bone density and increased susceptibility to fractures, is a bone disease that often corresponds with the decrease of sex hormones within the body. It is today’s most prevalent metabolic bone disease (Mills et al., 2021). The relationship between sex hormones, primarily estrogen and testosterone, and osteoporosis is just one aspect of bone health that unfolds across our lifespan, particularly during menopause for women and later life for men and women.
Estrogen, an essential sex hormone primarily produced by the ovaries in females, plays a central role in maintaining bone density. During the reproductive years, estrogen is essential to the production of osteoblasts, the cells responsible for bone formation. It promotes bone matrix synthesis and inhibits osteoclast activity, the cells responsible for bone resorption. The balance between bone formation and resorption helps ensure optimal bone density. The relationship between estrogen and bone health becomes particularly evident during menopause, the physiological stage marking the end of a woman’s reproductive capacity. As women transition through menopause, there is a significant decline in estrogen production. This hormonal shift disrupts the equilibrium between bone formation and resorption, causing more bone to break down (if estrogen promotes bone formation and is decreasing, resorption will naturally become more prevalent.) Accelerated bone resorption reduces bone density, rendering postmenopausal women more liable to fractures, especially in weight-bearing bones. Estrogen’s influence even extends beyond its direct effects on bone cells. Estrogen plays a role in maintaining calcium balance, a crucial component of bone mineralization. Reduced estrogen levels lead to calcium imbalance, compromising our bones’ health. Interaction between estrogen and other hormones, such as parathyroid hormone and vitamin D, further defines the complexity of hormonal regulation and skeletal homeostasis.
While estrogen takes center stage in women’s bone health, testosterone, the primary male sex hormone, contributes significantly to bone health in men. Produced predominantly in the testes, it stimulates the growth and strengthening of bones, especially during puberty and adolescence. It promotes the development of peak bone mass, a critical determinant of bone health in late life. In men, the decline in testosterone levels with aging can contribute to bone loss, albeit to a lesser extent than observed in women during menopause. The gradual reduction of testosterone production is associated with increased bone resorption and decreased bone mineral density. Furthermore, conditions that result in reduced testosterone levels, such as hypogonadism, can exacerbate the risk of osteoporosis in men. Expanding on possible conditions, the following all contribute to reducing hormone circulation in both men and/or women: primary ovarian insufficiency, hypothalamic amenorrhea, congenital hypogonadotropic, pregnancy and lactation-associated osteoporosis, and hyperprolactinemia. All of these are just a few of the many examples highlighting the importance of sex hormones on our skeletal homeostasis, as they all in some way contribute to bone mineral density (Mills et al., 2021)
Osteoporosis is essentially a consequence of hormonal imbalance disrupting the balance between osteoblast and osteoclast activity. The decline in sex hormones in both men and women leads to increased bone resorption. This imbalance compromises the overall bone structure, making it more porous and prone to fractures. The impact of hormonal changes on bone health is not limited to the structural aspect but also extends to their mechanical properties. Bone density affects bone strength, making individuals more susceptible to fractures even with minimal trauma. Spinal compression fractures, hip fractures, and wrist fractures become more common occurrences while also proving to be fatal.
Even with as daunting as age’s effects seem on our bones, there are a few steps we can take to help manage/prevent osteoporosis. For one, hormone replacement therapy (HRT), which involves supplementing estrogen in postmenopausal women, has been a longstanding approach to reducing a hormonal decline’s effect on our bone’s health (Almeida et al., 2017). HRT aims to restore hormonal balance, reduce bone resorption, and minimize bone fracture risk. The way we choose to live our lives can also play a part in promoting maximum gone health for ourselves. Between adequate intake of vitamin D and calcium, as well as consistent weight-bearing exercise, these three things combined represent the best ways to keep our bones healthy. In fact, vitamin D has been found to promote testosterone and sperm production in males, which, if you remember, promotes osteoblast activity (Mills et al., 2021). Experimental studies support a beneficial effect of vitamin D on male fertility, by modulating hormone production through genomic and non-genomic action (de Angelis et al., 2017). This improvement in male hormone production is obviously going to stimulate osteoblasts, leading to an increase in bone formation. Smoking cessation and moderation in alcohol consumption are additional lifestyle modifications that can improve our bone health for the better (Endocrine Society, Martinez).
As can be seen, the relationship between sex hormones and osteoporosis is more than evident. Estrogen and testosterone, the primary sex hormones in women and men, respectively, play the most significant roles in ensuring our bones are optimally healthy. The decline in these hormones, most notably during menopause and late-life aging, disrupts the balance between bone formation and resorption, predisposing individuals to osteoporosis. However, by recognizing the significance of our hormones’ role in bone health, we can enhance our understanding of osteoporosis and better help ourselves prevent/control its effects!
REFERENCES:
Mills, E. G., Yang, L., Nielsen, M. F., Kassem, M., Dhillo, W. S., & Comninos, A. N. (2021). The Relationship Between Bone and Reproductive Hormones Beyond Estrogens and Androgens. Endocrine Reviews, 42(6), 691–719. https://doi.org/10.1210/endrev/bnab015
Almeida, M., Laurent, M. R., Dubois, V., Claessens, F., O’Brien, C. A., Bouillon, R., Vanderschueren, D., & Manolagas, S. C. (2017). Estrogens and Androgens in Skeletal Physiology and Pathophysiology. Physiological Reviews, 97(1), 135–187. https://doi.org/10.1152/physrev.00033.2015
de Angelis, C., Galdiero, M., Pivonello, C., Garifalos, F., Menafra, D., Cariati, F., Salzano, C., Galdiero, G., Piscopo, M., Vece, A., Colao, A., & Pivonello, R. (2017). The role of vitamin D in male fertility: A focus on the testis. Reviews in endocrine & metabolic disorders, 18(3), 285–305. https://doi.org/10.1007/s11154-017-9425-0
Martinez, R. E. (m.d.). An endocrine society patient resource hormones and your bones. https://www.endocrine.org/-/media/endocrine/files/patient-engagement/hormones-and-series/hormones_and_your_bones_what_you_need_to_know.pdf

Ethan’s project defines the roles of sex hormones, specifically estrogen and testosterone, in developing osteoporosis and maintaining bone health. It starts with outlining how bones develop from in utero stages through endochondral ossification, which is a process that’s heavily influenced by hormonal interactions. During puberty, higher levels of sex hormones lead to higher bone mass development, which is important before the natural decline of hormone levels that comes with age.
The focus on osteoporosis, which is a prevalent bone disease that is defined by decreased bone density and increased fracture risk. Osteoporosis is a direct result of diminished sex hormone levels. Estrogen, which is essential for women, facilitates bone formation and resorption, and maintains bone density. However, estrogens significant reduction during menopause leads to an imbalance in osteoclasts (bone remodeling/reabsorption cells), leading to bone loss. In men, testosterone promotes bone mass and strength, and its gradual decline also contributes to bone density loss, though its less significant than estrogen in women.
Ethan also goes over intervention strategies for osteoporosis, such as Hormone Replacement Therapy (HRT) for postmenopausal women, which attempts to restore hormonal balance and reduce bone loss. Many lifestyle factors like adequate intake of vitamin D and calcium, regular weight training, quitting smoking, and lower alcohol consumption are also possible preventative measures against osteoporosis.
The project demonstrates the essential role of sex hormones in bone health and the development of osteoporosis, which shows the importance of understanding these hormonal dynamics to help in preventing and managing this condition.