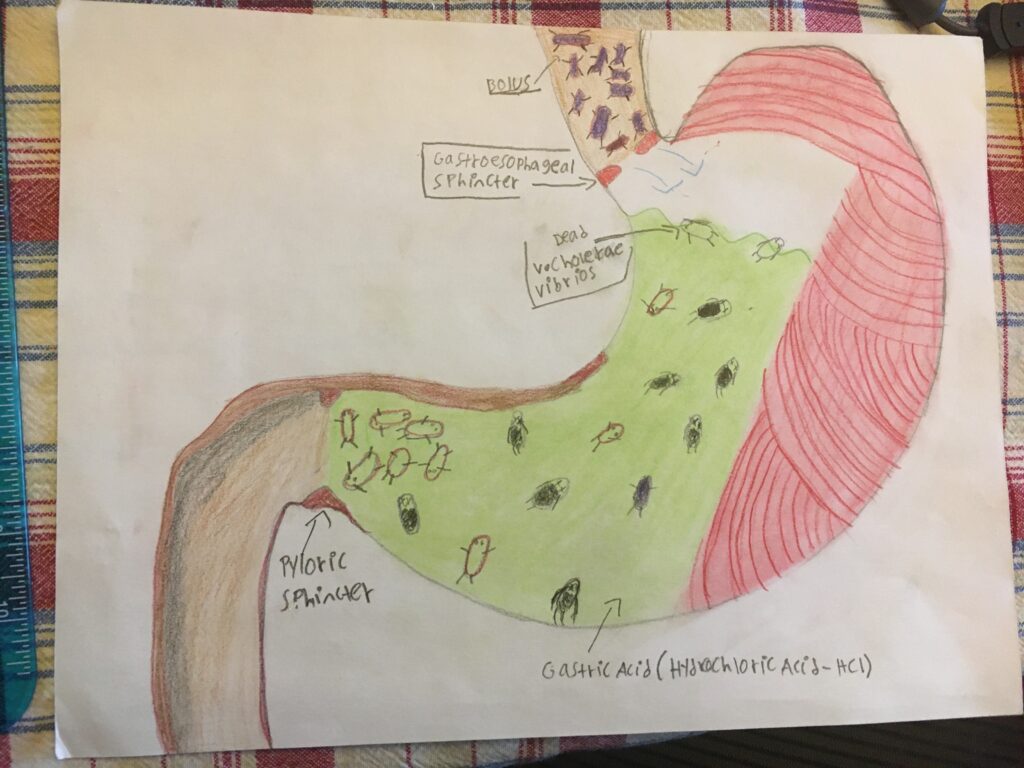
My project is about how Cholera attacks the Digestive System and cripples the Renal System.
I chose to illustrate the entire pathway of the V. Cholera Vibrios, then Cholera Toxin (CT), through the digestive and renal system. After ingestion of the V. Cholerae Vibrios (bacteria, they )face their biggest obstacle preventing an infection – the gastric acid (Hydrochloric Acid HCI). The majority if not all of V. Cholerae Vibrios will be killed while passing through the stomach, before they reach the small intestine. This is why a majority of people who are infected with the V. Cholera vibrios show mild to zero symptoms.
The left side of the second illustration are 2 divided sections of the small intestine, there farthermost left occurs after the Cholera Toxin (CT) ultimately results in the rapid secretion of water and electrolytes into the small intestine. The blue represents water as the volume levels are extremely high compared to normal levels as seen with the adjacent portion of the illustration. Additionally the amount of V. Cholerae has grown significantly larger than in the beginning as with the adjacent portion.
The right side of the illustration details the steps that the CT has to go through throughout the cell. The V. Cholerae Vibrios produce the CT when they reach the small intestine, along with Mucolytic enzymes, proteases (Protein digesting enzyme/factors), and Hemagglutinin (Binding factor for the B unit to the GM1 Gangliosides (Glycolipid) on the surface of the intestinal epithelial cells.
The CT is composed of 2 parts, the B unit, composed of 5 peptide chains, and the A group – composed of a A1 and A2 unit.
The first stage is characterized by the B unit binding/fusing itself to the GM1 Gangliosides (Glycolipid) on the plasma membrane. The 2nd part involves the A unit to enter the cell into the cytoplasm. Here it first goes through the Golgi Apparatus (GA), and then to the Endoplasmic Reticulum (ER). At this location the previously mentioned A1 subunit will sever the (disulfide) bond between the A2 and the B unt. The A2 unit will remove ADP from NAD+ and transport it to the GTP (G) protein. Here it will “increase” the stimulation of the Adenylate cyclase (AC). This will cause the AC to constantly be working/producing Cyclic Adenosine Monophosphate (cAMP). This stimulates the Protein Kinase A (PKA) which in turn stimulates the Cystic Fibrosis Transmembrane Ion Channel. With the channel now being opened all the time, there is a massive release of water, bicarbonate (HCO3-) and electrolytes such as chloride. This results in acute severe diarrhea and heavy electrolyte imbalance/deficiency. If medical treatment is sought immediately, the disease is no longer life threatening as the best treatment is rapid rehydration either orally or intravenously.
The V. Cholerae vibrios transfer into a temporary hyper infectious (HI) state post shedding by the infected host into the wild. This time period can range from 5-10 hours, depending on the amount/volume of already present V. Cholera Vibrios within the present environment – initiation competition, which reduces the general HI timespan. This information has been used to explain the reasoning behind the explosive nature of cholera outbreaks/epidemics, and how they are able to spread so quickly within a short period of time.

- Cholera NIH) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3761070/ -[Harris, J. B., LaRocque, R. C., Qadri, F., Ryan, E. T., & Calderwood, S. B. (2012). Cholera. Lancet (London, England), 379(9835), 2466–2476. https://doi.org/10.1016/S0140-6736(12)60436-X]
Cholera. (2022, March 30). World Health Organization. Retrieved April 21, 2023, from
Cholera. (2019, December 12). Mayo Clinic. Retrieved April 21, 2023, from https://www.mayoclinic.org/
diseases-conditions/cholera/symptoms-causes/syc-20355287
- NIH Pathogenesis Of Cholera) https://www.ncbi.nlm.nih.gov/books/NBK8407/
Finkelstein RA. Cholera, Vibrio cholerae O1 and O139, and Other Pathogenic Vibrios. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 24. Available from: https://www.ncbi.nlm.nih.gov/books/NBK8407/
- Hemagglutinin-Neuraminidase) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC136693/
Takimoto, T., Taylor, G. L., Connaris, H. C., Crennell, S. J., & Portner, A. (2002). Role of the hemagglutinin-neuraminidase protein in the mechanism of paramyxovirus-cell membrane fusion. Journal of virology, 76(24), 13028–13033. https://doi.org/10.1128/jvi.76.24.13028-13033.2002
Davy Vanden Broeck, Caroline Horvath, Marc J.S. De Wolf,
Vibrio cholerae: Cholera toxin,
The International Journal of Biochemistry & Cell Biology,
Volume 39, Issue 10,
2007,
Pages 1771-1775,
ISSN 1357-2725,
(https://www.sciencedirect.com/science/article/pii/S1357272507002208)
Abstract: The bacterial protein toxin of Vibrio cholerae, cholera toxin, is a major agent involved in severe diarrhoeal disease. Cholera toxin is a member of the AB toxin family and is composed of a catalytically active heterodimeric A-subunit linked with a homopentameric B-subunit. Upon binding to its receptor, GM01, cholera toxin is internalized and transported in a retrograde manner through the Golgi to the ER, where it is retrotranslocated to the cytosol. Here, cholera toxin reaches its intracellular target, the basolaterally located adenylate cyclase which becomes constitutively activated after toxin-induced mono-ADP-ribosylation of the regulating GS-protein. Elevated intracellular cAMP levels provoke loss of water and electrolytes which is manifested as the typical diarrhoea. The cholera toxin B-subunit displays the capacity to fortify immune responses to certain antigens, to act as a carrier and to be competent in inducing immunological tolerance. These unique features make cholera toxin a promising tool for immunologists.
Keywords: Cholera toxin; Vibrio cholerae; Retrograde traffic; Immunological adjuvant
- Acute Tubular Necrosis) https://www.ncbi.nlm.nih.gov/books/NBK507815/
Hanif MO, Bali A, Ramphul K. Acute Renal Tubular Necrosis. [Updated 2023 Feb 19]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK507815/
- Pathogenesis Of Cholera – The Lancet https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)00330-0/fulltext#seccestitle40
[Kanungo, Azman, https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)00330-0/fulltext#seccestitle40]
[Lingwood, Mafoud, Microbial Glycobiology Structures, Relevance and Applications]
AUTHOR=Ramamurthy Thandavarayan, Nandy Ranjan K., Mukhopadhyay Asish K., Dutta Shanta, Mutreja Ankur, Okamoto Keinosuke, Miyoshi Shin-Ichi, Nair G. Balakrish, Ghosh Amit
TITLE=Virulence Regulation and Innate Host Response in the Pathogenicity of Vibrio cholerae
JOURNAL=Frontiers in Cellular and Infection Microbiology
VOLUME=10
YEAR=2020
URL=https://www.frontiersin.org/articles/10.3389/fcimb.2020.572096
DOI=10.3389/fcimb.2020.572096
ISSN=2235-2988
ABSTRACT=The human pathogen Vibrio cholerae is the causative agent of severe diarrheal disease known as cholera. Of the more than 200 “O” serogroups of this pathogen, O1 and O139 cause cholera outbreaks and epidemics. The rest of the serogroups, collectively known as non-O1/non-O139 cause sporadic moderate or mild diarrhea and also systemic infections. Pathogenic V. cholerae circulates between nutrient-rich human gut and nutrient-deprived aquatic environment. As an autochthonous bacterium in the environment and as a human pathogen, V. cholerae maintains its survival and proliferation in these two niches. Growth in the gastrointestinal tract involves expression of several genes that provide bacterial resistance against host factors. An intricate regulatory program involving extracellular signaling inputs is also controlling this function. On the other hand, the ability to store carbon as glycogen facilitates bacterial fitness in the aquatic environment. To initiate the infection, V. cholerae must colonize the small intestine after successfully passing through the acid barrier in the stomach and survive in the presence of bile and antimicrobial peptides in the intestinal lumen and mucus, respectively. In V. cholerae, virulence is a multilocus phenomenon with a large functionally associated network. More than 200 proteins have been identified that are functionally linked to the virulence-associated genes of the pathogen. Several of these genes have a role to play in virulence and/or in functions that have importance in the human host or the environment. A total of 524 genes are differentially expressed in classical and El Tor strains, the two biotypes of V. cholerae serogroup O1. Within the host, many immune and biological factors are able to induce genes that are responsible for survival, colonization, and virulence. The innate host immune response to V. cholerae infection includes activation of several immune protein complexes, receptor-mediated signaling pathways, and other bactericidal proteins. This article presents an overview of regulation of important virulence factors in V. cholerae and host response in the context of pathogenesis.
- Hyper Infectivity Sate (HI) of Cholera https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1298942/
Hartley, D. M., Morris, J. G., Jr, & Smith, D. L. (2006). Hyperinfectivity: a critical element in the ability of V. cholerae to cause epidemics?. PLoS medicine, 3(1), e7. https://doi.org/10.1371/journal.pmed.0030007
- Peer Reviewed Hyper Infectious state of V. Cholerae (HHS) https://www.nature.com/articles/nrmicro2204
Nelson, E., Harris, J., Glenn Morris, J. et al. Cholera transmission: the host, pathogen and bacteriophage dynamic. Nat Rev Microbiol 7, 693–702 (2009). https://doi.org/10.1038/nrmicro2204

This project described the impact of cholera attacks on the digestive system and the resulting toxic impact on the renal system. This relates to the objective, “describe the function of electrolytes and solutes and their regulation”. If Cholera Toxin (CT) is not killed by acid in the stomach, the bacterium travel to the small intestine. This causes the Cystic Fibrosis Transmembrane Ion Channel. to remain open constantly allowing significant amounts of fluid and electrolytes to flow out freely resulting in electrolyte imbalance/deficiency and dehydration due to acute severe diarrhea. Without proper and immediate rehydration the disease can be deadly due to kidney failure in response to an extreme and rapid loss of hydration and electrolytes.