STEAM Project
Topic: Analyzing the process of how broken bones heal.
Objective: Know the stages of bone development and repair.
Bone Development:
Most people when they think of bones imagine a hard, white skeleton that is only useful because it helps the body maintain an erect posture, protects important organs, and makes a great Halloween costume (Anatomy; 6.1, pg. 214). While this is all true, bones that comprise our skeletal system are more complicated than what people think. For example, the development of bones provides a clear insight into the intricacies of this connective tissue and how it assists our body with many functions.
The formation of bone begins in the early stages of embryonic development when the skeletal structure is only hyaline cartilage and fibrous membranes. As the embryo matures into the ripe old age of about six or seven weeks, the process known as ossification, osteogenesis, starts (Anatomy; 6.4 pg. 231). Nevertheless, it is important to note that ossification is the process of bone formation. In this case, the cartilage is gradually becoming replaced with bone, so this development is known as endochondral ossification. This procedure is made possible through osteoblasts; a connective tissue that secretes osteoids which is a “gelatinous substance made up of collagen, a fibrous protein, and mucopolysaccharide, an organic glue.” After this secretion, a deposit of inorganic salts hardens and becomes bone. This eventually causes dying cartilage cells which becomes replaced with ossification centers which is where bone formation derives from (Britannica). Overtime, endochondral ossification continues throughout childhood but still leaves a little for adulthood.
Art Project of Bones Repairing:
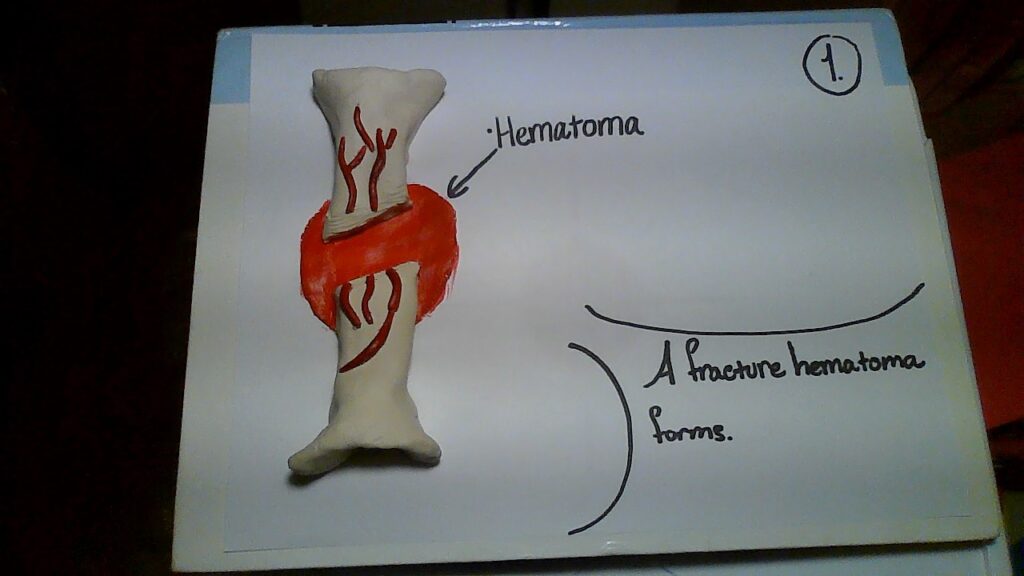
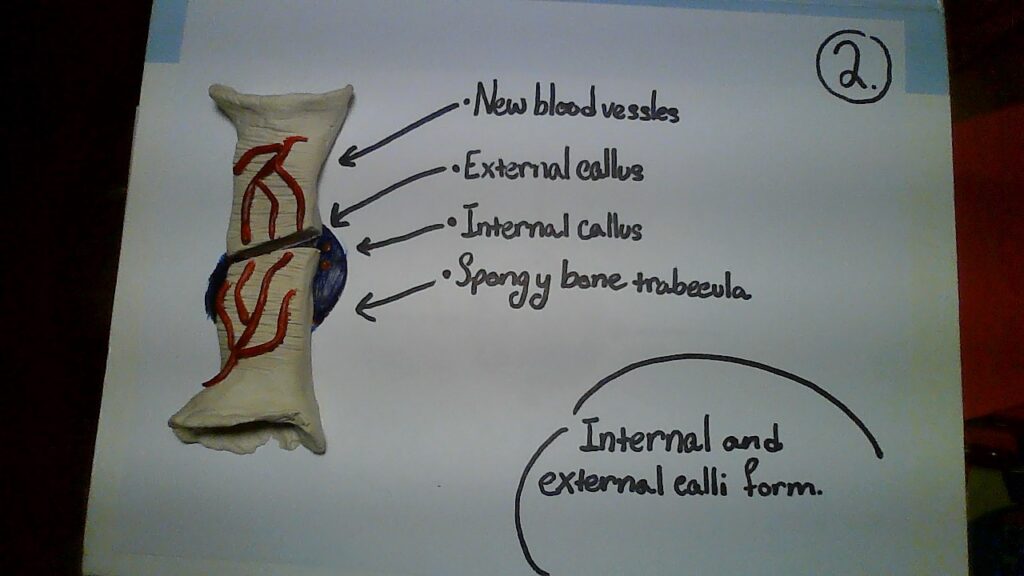
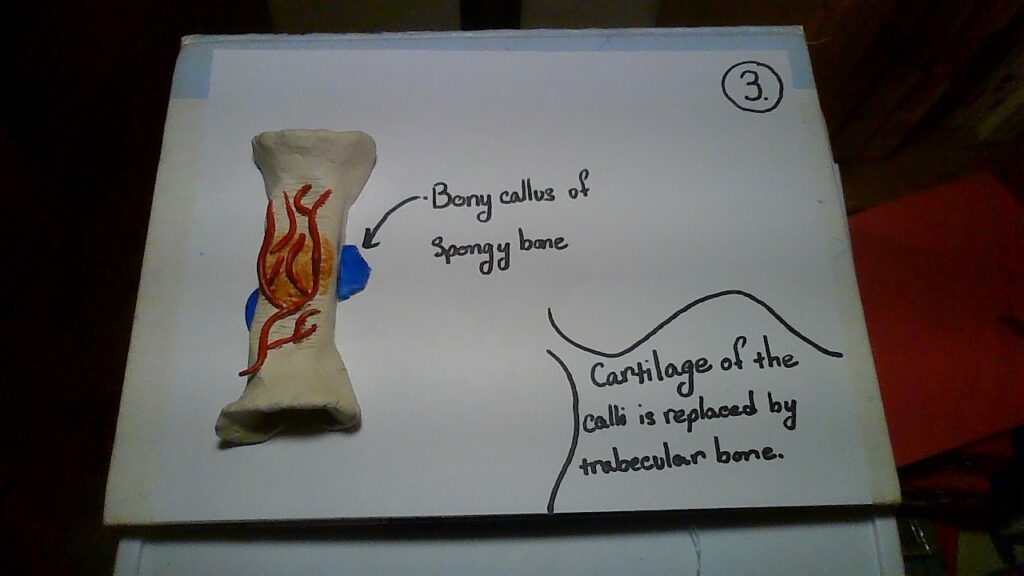
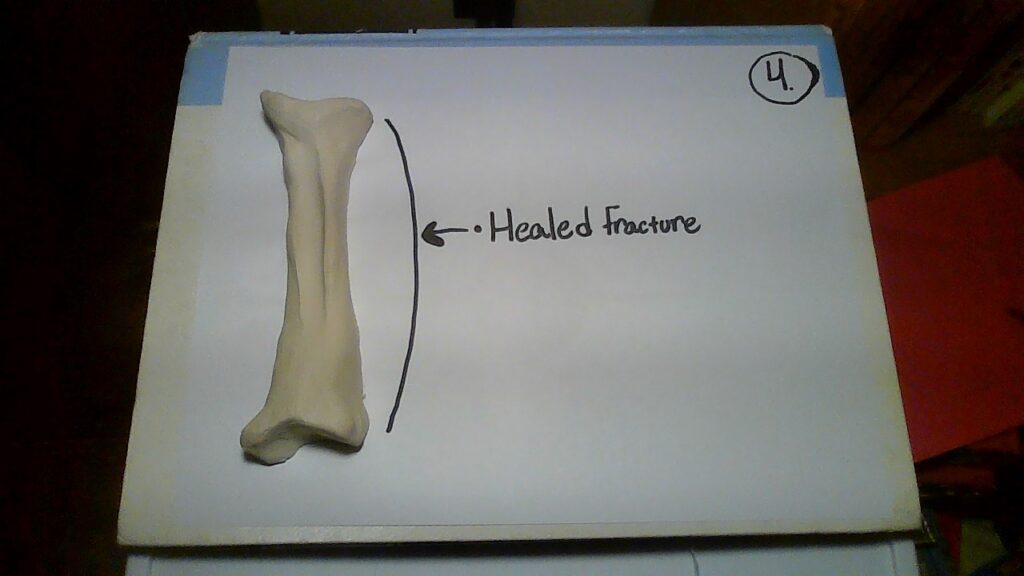
In the pictures, it shows the process of bone repair through air dry clay models and acrylic paint. Although there are many specific types of fractures, the pictures show a basic outline to provide readers with a visual of broken bones.
- The fracture of a hematoma is formed as a result of a break.
- The internal and external calli becomes organized.
- The trabecular bone replaces the cartilage of the calli.
- Lastly, the remodeling of the bone happens, and the fraction becomes healed.
Nevertheless, this complicated process begins after the breakage of a bone when blood flows from the torn vessels and begins to clot. As a result of this, about 6-8 hours after the breakage, the clot becomes a fractured hematoma. Sadly, many cells surrounding the fracture die because of blood flow disruption. Next, an internal callus devises chondrocytes that come from the endosteum; this is caused at the end of two broken bones by the secretion of fibrocartilaginous matrix. Adding on to that, an external callus, composed of bone hyaline cartilage, is made from osteoblasts and chondrocytes. After this stabilization, the dead bone becomes resorbed through osteoclasts and osteogenic cells divide into osteoblasts. As time goes by, the spongy bones become replaced by the compact bone and the calli unite. Overall, after this long process of several weeks, the bone is healed, and the visible exterior of our arm indicates no remains (Anatomy; 6.5, pg. 240).
Bone Repair:
“Sticks and stones may break my bones” but I know my body can heal. Intriguingly, a broken bone is a fracture that involves damaged tissues, and the specific category of the breakage varies on the amount of injury. Nevertheless, a person can tell that it is fractured because the ruptured blood vessels, injured muscles and ligaments, or torn skin. However, the ability to repair itself is a unique characteristic of bone and this process is made possible through the release of mediators. Moreover, the effects of the mediators can be observed as some scientists have observed:
The first event is immediate and consists of the activation of the coagulation cascade and the formation of a blood clot. Shortly afterward there follows an acute inflammatory response resulting in tissue edema and cytokine and growth factor release. Then follows the first stage of collagen repair, involving deposition and the formation of granulation tissue, which becomes a new and temporary weak tissue. The third and final process is the second phase of collagen repair, resulting in extracellular matrix remodeling, angiogenesis, and the reproduction of full-strength tissue. Much of the normal healing process is driven by growth factors and cytokines (Braddock).
In other words, after the breaking of a bone, the process of healing is made possible through growth factors and cytokines.
Additionally, researchers have found that through the help of stem cells, the bone can recover. For instance, stem cells assist tissues in regenerating and renewing themselves. Furthermore, stem cells can become blood vessels, cartilage or bone depending on the medium and treatment for the problem; moreover, the ability to regenerate more of the above components are ideal in the healing process of bones (Ebraheim). Adding to that fact, although the impacted damage of a bone will vary, the repair requires the deposition of mineralized matrix through the assistance of stem cells. The stem cell sources mainly come from periosteum and bone marrow. However, there are two specific types of stem cells that assist in bone repair: periosteal and BMSCs cells. The periosteum cell is a source for osteoblasts and chondrocytes while the BMSC cells contribute to the chondrocytes but not as much in comparison to the periosteum cells (Ono and Kronenberg).
References Cited
Betts, Gordan J. Young, Kelly A. Wise, James A. (2017). Anatomy and Physiology. Houston, Texas: Rice University.
Braddock, Martin. Houston, Parul. Campbell, Callum. Ashcroft, Patrick. (2001). “Born Again Bone; Tissue Engineering for Bone Repair.” American Physiology Society, 16:5. 208-213. https://journals.physiology.org/doi/epdf/10.1152/physiologyonline.2001.16.5.208
Britannica, T. Editors of Encyclopedia (2020, May 28). “Bone Formation.” Encyclopedia Britannica. https://www.britannica.com/science/bone-formation
Ebraheim, Nabil. (2011). “Stem Cells and Bone Healing – Everything You Need to Know.” YouTube. https://www.youtube.com/watch?v=w9tWsEzsPkk
Ono, N., & Kronenberg, H. M. (2016). “Bone Repair and Stem Cells.” Current opinion in genetics & development, 40, 103–107. https://doi.org/10.1016/j.gde.2016.06.012
